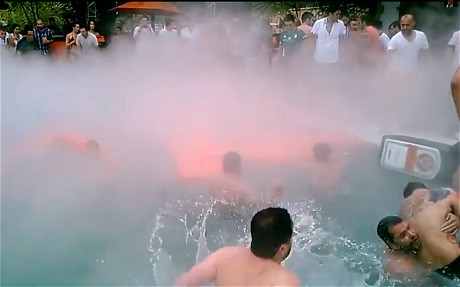Poison Gas at Disneyland Paris | What kind of a vacation is this?!
 The last thing I expect to encounter on vacation is poison gas at Disneyland Paris. Apparently someone, who needs to revisit the training manual, combined ammonia with sodium hypochlorite (bleach) to create a deadly gas and coincidentally nearly asphyxiate several visitors to le Royaume magique.
The last thing I expect to encounter on vacation is poison gas at Disneyland Paris. Apparently someone, who needs to revisit the training manual, combined ammonia with sodium hypochlorite (bleach) to create a deadly gas and coincidentally nearly asphyxiate several visitors to le Royaume magique.
According to news reports, 22 people were treated for a variety of respiratory complaints after several chemicals were incorrectly combined and dumped into the pool at the New York hotel in Disneyland Paris. Many of the news reports state the offending agent was potentially lethal chloramine gas.
Poison Gas at Disneyland Paris?! Say what?!
The chloramines are a family of chemicals produced when the hydrogen in ammonia is replaced with chlorine; monochloramine (chloroamine, NH2Cl), dichloramine (NHCl2), and nitrogen trichloride (NCl3). Organic chemicals can be formed by adding carbon molecules to the nitrogen of either monochloramine or dichloramine. It’s not all that uncommon to develop a chloramine vapor cloud over a swimming pool because the chloramine rises out of the water and since its heavier than air, settles over the surface of the pool water. That’s very often the “chlorine” smell that you encounter when you enter a poorly ventilated indoor pool.
Several news outlets report that the poison gas at Disneyland Paris was a chloramine gas cloud created when sulfuric acid was mixed with bleach and this may be true – as we know from a previous blog post, bleach is sodium hypochlorite. Household bleach is typically 3-5% sodium hypochlorite. The “liquid chlorine” you buy at the pool store is generally 10-12% sodium hypochlorite. So there is plenty of high concentration bleach available around the pool.
In America we tend to use muriatic acid more often in our pools to lower the pH. In Europe and in particular Germany and France they use sulfuric acid for the same purpose. Sulfuric acid (H2SO4) is a colorless, odorless and viscous liquid. Sulfuric acid is an extremely corrosive oxidizer. When diluted in a water solution, sulfuric acid dissociates into a hydrogen (H+) cation and sulfate (SO4-2) anion. Sulfuric acid in water creates a highly acidic solution with a pH that varies according to the proportion of sulfuric acid to water. When an acid is mixed with sodium hypochlorite, the acid will donate a hydrogen molecule to the compound, replacing the sodium molecule (Na) to produce hypochlorous acid (HClO). Mixing sulfuric acid with a solution of sodium hypochlorite will result in a solution of sodium sulfate (Na2SO4) and hypochlorous acid.
The reaction of sulfuric acid and sodium hypochlorite, which was likely the scenario for the poison gas at Disneyland Paris, doesn’t stop with the production of sodium sulfate and hypochlorous acid. In water solutions (like a pool), hypocholorite (HClO) and chlorine (Cl2) reach an equilibrium that is dependent on the pH of the solution. In an acidic solution (the more sulfuric acid you add, the more acidic the solution becomes), the equilibrium favors chlorine in the following fashion: Hypochlorous acid partially breaks down into the hypochlorite anion and hydrogen cation. Hypochlorous acid is a strong oxidant, so the remaining hypochlorous acid in the solution oxidizes the hypochlorite anion producing the irritating and toxic chlorine gas (Cl2).
So what likely happened is that someone added too much chlorine to the ammonia and released a cloud of chlorine gas, which is a mucous membrane irritant. It’s not a terribly effective suffocating agent or chemical warfare agent because people realize they are being exposed to the chemical and they get out of the area really quickly, usually with tearing eyes, coughing and choking.
Perhaps instead of the chemicals we put in pools, we ought to really be concerned with what other people are putting in pools. There was a great study published last year out of Canada in which a team of researchers tested the water in several dozen swimming pools and found that 31 of the 31 swimming pools the team tested contained an artificial sweetener that could have gotten there only through people peeing it out in the pool.
acs.estlett.7b00043Urine, along with body care products like shampoos, lotions and conditioners, interacts with chlorine, ammonia, boric acid, bleach and other pool-sterilizing chemicals to form volatile organic compounds that can be unhealthy to breathe.
Yeesh.

Still on vacation at the beach – think I’m in love with the chicken fried chicken at Cracker Barrel.